 
A person can access the eMed service through Abbott's NAVICA app – available in English and Spanish – which can be downloaded from the Apple and Android app stores.Abbott is expanding further so more people have access to the tests. Those test are currently being distributed through the federal government. Since launching BinaxNOW in August, Abbott has ramped up capacity to 50 million tests a month in its U.S. "That’s why Abbott is bringing our rapid BinaxNOW test and NAVICA platform into homes through this partnership with eMed, which allows us to maintain the integrity of the testing process, get even closer to people who need testing and help provide the confidence we need to help get back to living with a bit more normalcy."Ībbott and eMed expect to deliver and administer 30 million BinaxNOW tests in the first quarter of 2021, with an additional 90 million in the second quarter. Ford, Abbott’s president and chief executive officer. Unfortunately, we’re still hearing that many people can’t access testing as quickly as they need it," said Robert B. "As the pandemic has evolved, the need for rapid testing has only grown. The new EUA comes just three months after our original launch of BinaxNOW and it furthers our vision – to decentralize and democratize testing – by bringing testing to more people in more places to help slow the spread of the virus. And our test, coupled with the NAVICA app, gives proof of a valid test result through a digitally-authenticated NAVICA pass and helps preserve the integrity of reporting so that public health officials get critical, real-time data on the disease and how it’s spreading during the pandemic. Costing $25 through eMed (plus shipping), BinaxNOW is the lowest available price for an at-home COVID-19 testing service. Just as important, our home test will be affordable, accessible and ensures confidence in the home-testing process. It's a significant advancement from the other testing options where individuals collect their own samples without an expert’s guidance and then package the specimen, mail it to a lab and wait a couple days, or more, for results. The BinaxNOW test, which provides results in minutes and detects the virus in the early part of the disease when people are most infectious, will be used with an online service, provided by eMed, which helps guide people through the testing process. 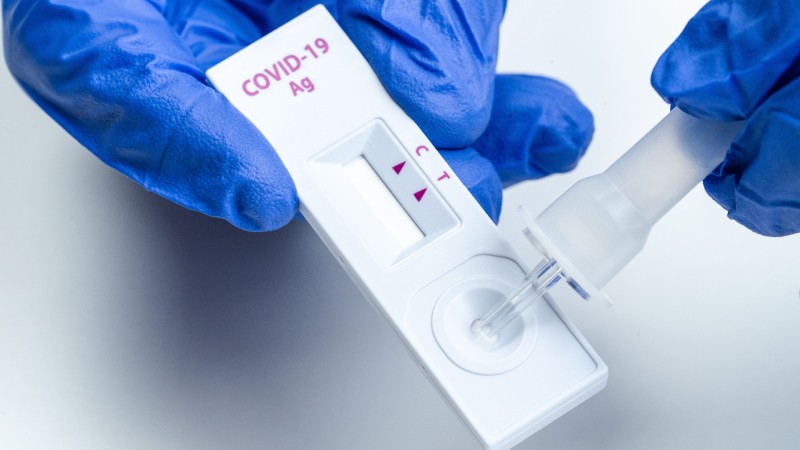
Food and Drug Administration (FDA) for use at home with a prescription through a virtually guided online service. Today we’re pleased to say that our widely-used BinaxNOW rapid test, the size of a credit card and requiring no instrumentation, has received emergency use authorization (EUA) from the U.S. We believe COVID testing should be more widely available, in more places, where people work, live, and study. We recommend checking with your airline and the local government of the country where you're traveling to confirm COVID-19 guidelines and testing requirements. As more parts of the world reopen and we co-exist with COVID-19, fast and reliable rapid tests will remain a critical tool. government has removed the requirement of a negative COVID-19 result to return to the U.S. Editor's Note: As of June 13, 2022, the U.S. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
